On the evening of July 1, 2014, Carolyn “Suzy” Markland, a 58-year-old Jacksonville, Florida, resident with a degenerative disc disease, took her prescribed medicine — a 400-microgram dose of a Fentanyl spray called Subsys — and went straight to bed.
Despite the fact that she regularly experienced pain, taking Subsys was not an everyday affair for Markland. Her prescription had been filled several months prior but she almost never took the stuff; her longtime family doctor and pharmacist had expressed to her plenty of no-holds-barred skepticism about it. On the three occasions she had taken Subsys, her family noticed that its sedative and respiratory effects were noticeably sharper than those of another strong painkiller she took, Exalgo.
On July 2, Markland visited Dr. Orlando Florete, her pain-management physician of five years, for a scheduled injection for her lower spine. As part of her anesthesia mix prior to the procedure, she received another Fentanyl dose. Unlike what was the case after previous procedures, however, she wasn’t up and moving some 20 to 30 minutes afterward; this time it took about an hour until her oxygen levels allowed for her to be safely released.
Markland was tired for the balance of the day and headed to bed early, skipping her usual cup of decaf beforehand.
She never woke up.
With Markland pronounced dead at 7:01 a.m. on July 3, the Jacksonville medical examiner’s office listed the cause of her death on its report as “drug toxicity,” noting the presence of Fentanyl and Exalgo. Her death was classified as “accidental.” The report also noted that Markland’s family doctor refused to sign the death certificate; Dr. Florete did.
Bob Markland, Carolyn’s husband of 19 years, declined to comment apart from providing a timeline of her Subsys use.
The medical examiner’s report of a lethal combination of Fentanyl and other drugs in Carolyn Markland’s blood is puzzling and sad, seemingly emblematic of a strain in modern American medicine whereby solutions to pain can be as scarce as the medication for that pain is abundant.
In another sense, this tale recounting Dr. Orlando Florete’s treatment presents a parallel trend in American medicine — that of the physician as a compensated endorser. According to figures from the Center for Medicare & Medicaid Services’ Open Payments database for the last five months of 2013, Florete was paid $18,874.03 by Subsys’ manufacturer to travel and speak to fellow doctors. The firm is a small but rapidly growing pharmaceutical company called Insys Therapeutics.
Additionally, the 16 Subsys prescriptions written by Dr. Florete from Jan. 1, 2013, to May 31, 2103, according to documents obtained by the Southern Investigative Reporting Foundation through the Freedom of Information Act, cost the U.S. military primary health insurance plan Tricare $133,770.36.
Pharmaceutical companies’ compensating physicians for discussing their product — or even attending carefully scripted seminars — is a longstanding, and legal, practice. To be certain, many within the medical community have been concerned about this for a while, and in 2013 regulations were put in place to ensure disclosure of all physician payments. (Pro Publica has published a wealth of information on the issue.)
A phone message seeking comment from Dr. Florete about his relationship with Insys and his Subsys prescription writing was not returned by the time of publication.
Like Dr. Florete’s speaking engagements, another unremarked-upon issue was the nature of Carolyn Markland’s Subsys prescription. The drug indicated to treat breakthrough cancer pain was prescribed for a bad back. The law affords doctors great latitude in determining whether drugs can be prescribed for reasons other than what they are designed for. On the other hand, doctors’ writing prescriptions based on off-label marketing have been at the center of nearly two dozen False Claims Act cases in the past 20 years, resulting in more than $13 billion in pharmaceutical company fines and settlement payments.
In the case of Subsys, its official label — indicated by the folded paper insert with the impossibly small typeface that comes with the package — notes that it’s contraindicated for those with headache pain and people not tolerant of the opioid class of drugs. According to the Centers for Disease Control and Prevention, 175,000 people died from some form of prescription opioid abuse from 1999 to 2010 compared with 120,000 from heroin and cocaine overdoses.
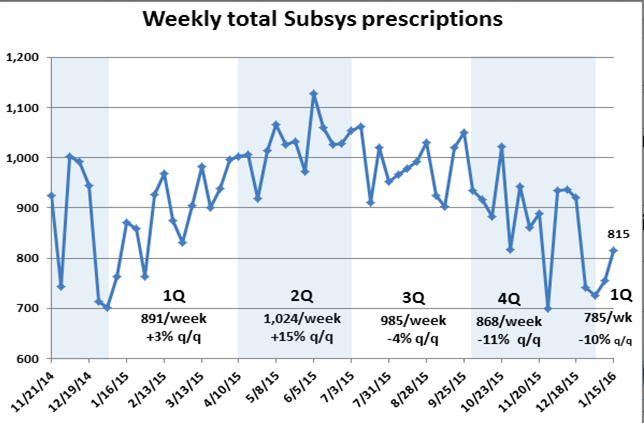
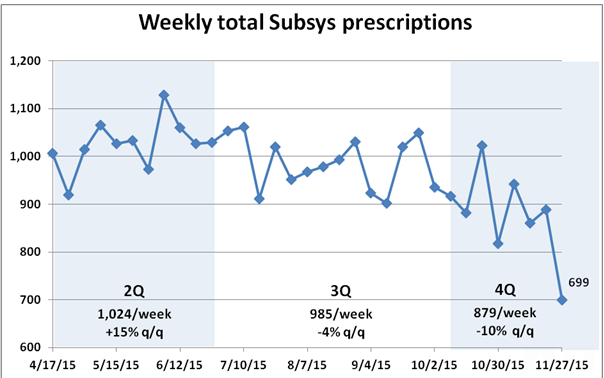
Like Dr. Florete, Insys Therapeutics has been doing pretty darn well. The company has had a remarkable level of financial success and its soaring stock price, as shown in the chart below, has made it a darling on Wall Street.

But that level of growth ought to warrant a raised eyebrow: Achieving in just two years more than $222 million in sales (from a level of about $15.5 million) without having invented something like a better search engine is no mean feat. Fentanyl, after all, has been around for many years. And while Subsys is the only spray version available, several Insys competitors are well-established and better capitalized and have sales forces that reach all 50 U.S. states.
While details about this breakthrough cancer pain medication are hard to find, or at least ones that are not self-serving management hype, veteran sales staff members from Insys and other pharmaceutical firms projected the company’s future growth rate to be roughly 10 percent a year. If this ends up being the case and the company is selling to oncologists, then the growth possibilities for Insys should be a function of that plus whatever business it can take away from its larger competitors. Many companies would be happy for those odds.
But Insys’ revenue grew north of 100 percent: Whatever organic growth the company is achieving is being aided by a whole lot of doctors who have grown profoundly fond of an expensive drug that’s accompanied by an acre of governmental red tape and one that the largest pharmacy benefit managers will no longer touch.
The question then becomes “how?”and “why?”
An investigation of Insys by the Southern Investigative Reporting Foundation reveals that this growth has come at a remarkable price: Food and Drug Administration data shows that Subsys is proving lethal to a growing number of patients, many of whom, like Carolyn Markland, are taking it for so-called off-label indications, such as headaches and back pain.
In reporting this story, the Southern Investigative Reporting Foundation repeatedly encountered former Insys employees who had received subpoenas requiring their appearance in front of a Department of Justice grand jury that has been empaneled in Boston. Still others had been interviewed for an investigation of the Department of Health and Human Services’ Office of the Inspector General.
A company that has been killing it — at least financially — is clearly in a lot of trouble.
————————
To understand Subsys, the first thing to know is that it is literally a drug apart: a Schedule II spray administered below the tongue and dozens of times stronger than morphine; its effects are profound, especially within the respiratory system, and almost immediately. Which is the point, of course, given that many people with cancer experience nausea and cannot take pills.
To address the twin risks of addiction and overdose, in March 2012 the Food and Drug Administration began what it calls the Transmucosal Immediate Release Fentanyl Risk Evaluation and Mitigation Strategy, blessedly shortened to TIRF-REMS. At its heart, the program is designed to make obtaining a prescription for Subsys (and five other drugs) a very deliberate process, with built-in checks and balances, such as confirmed opioid tolerance, signed patient statements and use of specially certified doctors and pharmacists.
No one, in other words, is dropping off a Subsys prescription at, say, a CVS pharmacy’s drive-through window.
Despite the unusual amount of federal guidelines designed to safeguard patients, Subsys is no stranger to adverse events.
The Southern Investigative Reporting Foundation asked Adverse Events, a California-based consultancy that collects and analyzes drug side effect data to analyze the FDA’s Adverse Event Reporting System’s tracking of fatalities related to Subsys. (In medical terms, an adverse event is defined as an undesirable outcome related to a drug’s use and includes categories in addition to death.)
The analysis shows Subsys was referenced in 63 adverse event reports resulting in deaths since its January 2012 FDA approval. Participation in the FAERS database is voluntary — a prescribing physician might not learn of an adverse event related to a drug; others elect not to report them. Because of this, many in the medical industry argue — privately — that FAERS’ data skews toward the lowest potential occurrence rate.
Given the relatively sparse amount of FAERS data that the Southern Investigative Reporting Foundation obtained (just age, gender and date of death are provided), placing the death of 63 Subsys users in a broader context is not so cut-and-dried. Certainly it’s reasonable to suppose that a percentage of those prescribed Subsys have cancer and would naturally have a higher rate of mortality. Some FAERS entries list Subsys along with one or two additional drugs. But dying of cancer isn’t usually considered an adverse pharmacological event; dying of respiratory failure when taking Subsys for a migraine is.
So how has Insys managed to grow exponentially?
The answer appears to have multiple parts: a truly unique sales force paired with a corporate speakers program that provides a stream of ready cash to frequent prescription writers.
————————
There’s no way around it: Insys’ sales force is very different from its competitors in the pharmaceutical industry. One reason is that a pharmaceutical sales background or even college courses in science are not required. Another is that if a candidate appears to be driven and aggressive, the company will look past things that a local Starbucks might not.
Scrolling through the LinkedIn profiles of Insys sales reps lends some credence to one of the assertions in an amended class action filed against the company in October that was settled the past week without a disclosure of the terms. The class action asserted that that Insys’ sales force was selected not for background or skill but for physical appeal
According to a summation of three confidential witnesses in the class action by the plantiffs’ lawyer, “most of Insys’s sales representatives were extremely attractive women.” (To be fair, Merck and other leading pharmaceutical companies have long drawn attention for constructing sales forces with a large percentage of attractive women.)
Then there’s the sales head of the New York region, Jeff Pearlman. Before becoming what his peers say is a highly productive salesman of Schedule II opioids, he was the marketing and sales chief of a company that sold aquariums.
Prior to that, he ran a ticket sales agency called Sitting Pretty Seating Services, which, in 2004, attracted the attention of the New Jersey Division of Consumer Affairs. Shortly afterward, the company’s registration was revoked after it did not file an annual report for two consecutive years, records indicate.
After this article’s initial posting, Pearlman said he had medical sales experience, having worked in the late 1990s for a company that sold diagnostic testing equipment to detect sleep apnea as well as for a company that sold genetic endocrinology testing devices in the mid-2000s.
Sunrise Lee, the recently departed head of Insy’s central and later Western sales region, offers an example of the company’s willingness to take a shot on a profoundly nontraditional prospect.
Prior to her stint with Insys, she was a dancer at Rachel’s, a West Palm Beach strip club. (She is the person at the far left top photo, taken from Rachel’s Web site, in this set; the bottom photo, from Facebook, shows a Insys sales outing at Chicago’s Wrigley Field for its top revenue producers.) It’s not clear what Lee did before adult entertainment.
About a year after Lee started selling one of the six drugs so lethal that the FDA had created a separate prescription protocol to monitor them, Insys promoted her to run the company’s Midwest sales.
SIRF asked Alec Burlakoff, Insys’ national sales chief, about the choice of Sunrise Lee to run sales for a quarter of the American land mass.
While agreeing with SIRF’s assertion that the adult entertainment world is not a traditional recruiting ground for pharmaceutical companies, Burlakoff offered that Lee had unusual attributes that were helpful in marketing Subsys to doctors.
“Doctors really enjoyed spending time with her and found Sunrise to be a great listener,” Burlakoff said.
“She’s more of a ‘closer,’” he said, using the common sales term often invoked to describe someone who helps convince a wavering customer to purchase a product. “Often the initial contact [with a doctor] was made by another sales person.”
SIRF asked Burlakoff about the scenario of a former exotic dancer pitching a restricted drug to board-certified oncologists. He said she was more effective with pain-management physicians who appreciated what he referred to as her “empathy.”
“When you are dealing with [doctors] who are around pain and cancer all day, an empathetic and caring sales person is helpful,” Burlakoff said. He said that Lee had been involved in an unnamed nutriceutical company prior to joining Insys and speculated that her “holisitic approach” to the medical field might also have appealed to some physicians. SIRF, having no idea what that means, asked him to elaborate; he did not. (SIRF couldn’t find or identify the company.)
For her part, Lee declined comment about Insys, noting that she had just been sued by the company — as was also the case for Lance Clark, an Insys sales executive from Dallas who had reported to her — for violating corporate policy regarding outside employment. The suit alleged that she recruited physicians to use a toxicology testing company, Advance Toxicology, that was formed by Clark when he was still employed by Insys. It also alleged that she made up having earned a degree from Michigan State.
She did however confirm to the Southern Investigative Reporting Foundation that she has been in contact with both the Department of Health and Human Services’ Office of the Inspector General and “those other prosecutors,” perhaps referring to the Department of Justice in Boston. (She declined to discuss it further when asked for clarification.)
Clark, who was unaware of the suit until the Southern Investigative Reporting Foundation told him about it, declined to comment.
————————
When asked about Insys’ controversial business practices, especially alleged off-label sales and payments made to physicians under its speakers bureau program (covered in New York Times investigations), Burlakoff insisted that these portrayals don’t match how he and his colleagues conduct themselves on a daily basis.
“There is a very, very easy way to get fired on your first day at this company,” said Burlakoff, “and that is to mention selling off-label. We are only selling a breakthrough cancer pain drug. That’s all we want to address with a doctor.”
“You don’t run a unit at a company like this by cutting corners,” he said. (Burlakoff was fired from Eli Lilly in 2003 for his role in sending unsolicited samples of Prozac through the mail in a bid to boost the drug’s then slumping sales. He and several colleagues sued the company, alleging management had approved of the plan.)
Having worked for rival drugmaker Cephalon, Burlakoff said he has run [Fentanyl] training programs “for years” and makes it clear to members of the sales staff that their job is not to try to convince doctors but educate them about the benefits and possibilities of a drug that can help their patients cope with a cancer-fighting regimen.
(The Department of Justice fined Cephalon $425 million in September 2008 for its off-label sales practices, particularly of its Fentanyl product, Actiq; Burlakoff is referenced in a qui tam complaint filed in 2014, for allegedly ordering his staff to organize speakers program events to promote off-label prescription of its Fentanyl drug. He did not respond to a request for comment about this via email and voice messages.)
The Southern Investigative Reporting Foundation asked Burlakoff about his previous assertion that the primary market for the drug was oncologists.
“Yes, well, we are trying to break in to that market but most [oncologists] only care about the tumor or malignancy and, in my opinion, don’t focus on the pain component,” he said. “That’s a problem — for them and for us.”
Adding that among oncologists there is a “sense that prescribing [Subsys] is something for hospice,” Burlakoff said most oncologists that he and his colleagues deal with are happy “to refer pain treatment out” to pain-management doctors so they could focus on the cancer treatment.
SIRF asked Burlakoff if the pain-management physicians who appear to be prescribing upward of 90 percent of the drug are thus working in tandem with oncologists or are otherwise treating cancer pain. He replied that this was his understanding based on what members of his sales staff were telling him.
“I can say that no one at Insys wants to see anyone taking [Subsys] for anything other than cancer pain,” said Burlakoff. He went on to relate several feel-good stories about people whose lives have been changed because of Subsys. More substantively, he referred to discussions he has had with Insys founder John Kapoor, whose wife Edith died of cancer in 2005, that motivate him to sell a product that eases the suffering of cancer patients.
————————
Also misunderstood, according to Burlakoff, was the role of Insys’ speakers program in the company’s sales model. It wasn’t, as the class action alleged via a confidential witness, “a kickback program.” Nor was it the way to incentivize a series of pain-management physicians to write more prescriptions, as a New York Times article suggested.
Rather, “putting board-certified doctors together, where one of them is explaining the benefits he or she is seeing” from prescribing Subsys is the way that the drug gets acceptance. No sales rep is as effective as a doctor at convincing other doctors, he said.
“These are rich, highly educated doctors,” Burlakoff said. “They have money. Whatever they are paid isn’t material.”
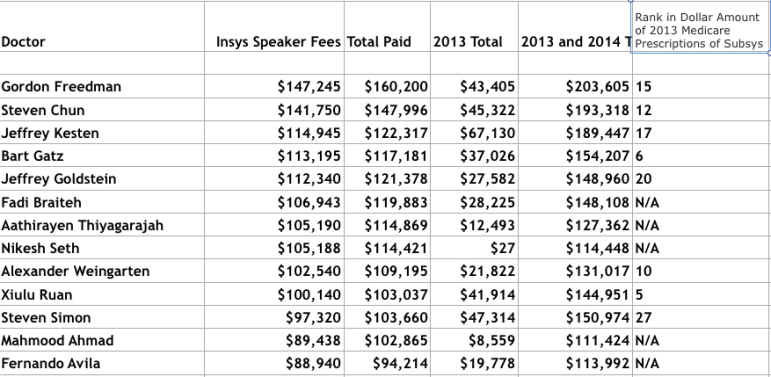
SIRF asked Burlakoff if money was not the primary motivation for the doctors whom Subsys paid $25,000, $50,000 or more over the last five months of 2013, then what did he suppose it was?
The chart below of the top nine recipients of Insys payments, drawn from the Center for Medicare & Medicaid Services’ Open Payments data, paints a clear picture of doctors who have generated substantial income from the program. (See a list of the top 25 recipients of Insys payments.) Burlakoff did not reply to a request for comment on this data.

Many of Burlakoff’s former colleagues, however, described a very different experience with the speakers program.
A qui tam claim filed last year by former Insys salesman Ray Furchak alleged that the speakers program’s sole purpose was, in the words of his then supervisor Alec Burlakoff, “to get money in the doctor’s pocket.” The catch, Furchak alleged, was that the doctors who increased the level of Subsys prescriptions, and at higher dosages (such 400 or 800 micrograms instead of 200 micrograms), would receive the invitations to the program — and the checks.
The claim described texts from Burlakoff to Furchak and other sales colleagues regularly demanding that “doctors be held accountable” and that “doctors who are not increasing their clinical experience [prescription writing], please cancel, suspend, and cease doing speaker programs.”
The Department of Justice chose not to join Furchak’s suit and he withdrew it. Reached at his new job, Furchak said he stood by everything he had alleged but declined to comment further.
Conversations with former sales staff members support Furchak’s allegations that the speakers program was regularly used as a lever to pressure doctors to increase dosage strength as well as the frequency of their prescriptions for Subsys. In return, former sales staff members (who were granted anonymity in this story because of their involvement with the Department of Justice’s grand jury proceedings) often had to deal with doctors’ annoyance about payment levels or delays in receiving their checks.
The speakers program events have often been held at branches of Roka Akor, a tony sushi-steak restaurant company with venues in Scottsdale, Chicago and San Francisco that’s owned by Insys founder John Kapoor. Based on interviews with multiple attendees, the expenses often run into the thousands of dollars and, given the sheer number of events, have helped his restaurants capture a handsome revenue stream. An email to Insys CEO Michael Babich seeking comment was not returned by the time of publication of this article.
Former sales staff members also disagreed that Burlakoff’s full-throated rejection of off-label sales was shared by upper management. As evidence of this, two former salespeople pointed to a quarterly meeting in Atlanta for the Southeast region sales team in a June 2014 when CEO Michael Babich, during a question and answer session, read a question about the risk of off-label sales, given Cephalon’s steep penalty in 2008.
“I understand why you’re asking that question,” said Babich. “But Cephalon didn’t have TIRF-REMS; we do. You are protected because both the MD and the patient have signed it.”
Asked to elaborate, Babich said because of the TIRF-REMS requirement that the patient be extensively briefed on the risks of Subsys, there couldn’t be a plausible claim that the patient (or doctor) did not know what he or she were doing.
As one of the two attendees who described this event to SIRF put it, “There wasn’t much else to say about the issue when your CEO sees an information protocol as an insurance policy.”
Putting Insys’ assertions about serving cancer patients aside, the company’s bread is buttered by pain-management and physical-rehabilitation doctors, according to Tricare’s reimbursement and prescription data from Jan. 1, 2013, to May 31, 2014. Tricare represents about 9.5 million people, or 3 percent of the U.S. population.
Listed below are Tricare’s top 15 prescribers of Subsys.

Among the top 25 Subsys prescribers within the Tricare system, there are 20 pain-management physicians, one osteopath, one nurse practioner and three physician assistants. (See a full list of the top 25.)
The Southern Investigative Reporting Foundation attempted to contact Dr. Xiulu Ruan and Dr. Patrick Couch, partners in a Mobile, Alabama, practice, about the fact that they were the leading Subsys prescription writers by an impressive margin, to discuss this, as well as their ownership of C&R Pharmacy, which dispenses the drug to their patients. (About 50 percent of the Subsys dispensed in the United States is handled by Linden Care, a specialty pharmacy on New York’s Long Island, owned by Bell Health Ventures, a private-equity fund.)
Anthony Hoffman, a lawyer representing the practice, told the Southern Investigative Reporting Foundation, “Based on your representation of the [Tricare] data you discussed with my client, we believe it to be inaccurate and encourage you not to publish it.” He did not specify what was wrong with the data and declined to provide further comment.
————————
As first reported in The New York Times, a series of Insys’ leading prescribers have been at the center of serious allegations involving their prescription-writing practices.
Last May federal prosecutors filed a complaint against Gavin Awerbuch, a Michigan-based pain-management physician and the company’s largest prescriber under Medicare (and third most compensated), for allegedly bilking Medicare out of $5 million over several years. Prosecutors allege that he wrote 20 percent of the Subsys prescriptions dispensed to Medicaid recipients nationwide from 2009 to 2014. (Subsys, however, has only been FDA-approved since January 2012.)
In December 2013 Judson Somerville, a Laredo, Texas-based pain-management physician (the No. 8 prescriber under Medicare and the most compensated) had his prescription-writing privileges “temporarily suspended” by the Texas Board of Medical Examiners for a host of findings, including having three patients die with six months of 2012; it was not the first time he had regulatory trouble.
Stewart Grote, a Lansing, Kansas, pain physician and the company’s fourth biggest Tricare prescriber (he received $8,48.05 from Insys), was sanctioned for multiple standard of care lapses and is no longer registered as a physician in that state, according to licensing records; he also had an earlier regulatory issue in 2010.
The Florida Department of Health sued Paul Wand and Miguel de la Garza, the No. 11 and 23 Tricare prescribers, in 2012. (Wand received $20,169.06 from Insys; de la Garza $17,019.04.) The department alleged Wand’s standard of care did not meet professional standards for a series of patients, particularly with regard to his prescription writing. With respect to de la Garza, the department claimed he did not professionally administer care to one specific patient. According to the Florida Board of Medicine’s Web site, both cases appear to be ongoing.
Chicago-based pain-management physician Paul Madison is not among the top 25 Tricare prescribers but he was the 17th most compensated under the speakers program. He was indicted in 2012 in connection with an alleged $3.5 million false insurance billings scam. The case is ongoing.
Heather Alfonso, the 25th largest Tricare prescriber of Subsys and a Derby, Conn.-based nurse practitioner, surrendered her state and federal nursing and prescription-writing licenses within the past month amid a Connecticut Department of Public Health investigation into her conduct. A February Connecticut Health I-Team story reported that in 2012, the most recent year for which data was available, she was among the nation’s top 10 prescribers of Schedule II substances within Medicare’s drug program.
The Southern Investigative Reporting Foundation asked CEO Michael Babich for comment via a detailed voice message left on his office phone and a pair of emails. He did not reply by publication time.
Clarification: This piece has been updated to clarify the description of former work roles of Jeff Pearlman, Insys’ New York regional sales manager. He served as the sales and marketing chief of an aquarium company. He also worked at two medical technology companies.
Update: This story was updated on March 22, 2016.